Advancing the a posteriori quest for deep-blue phosphorescence: Quantifying excitation-induced metal-to-ligand charge transfer as a guiding indicator
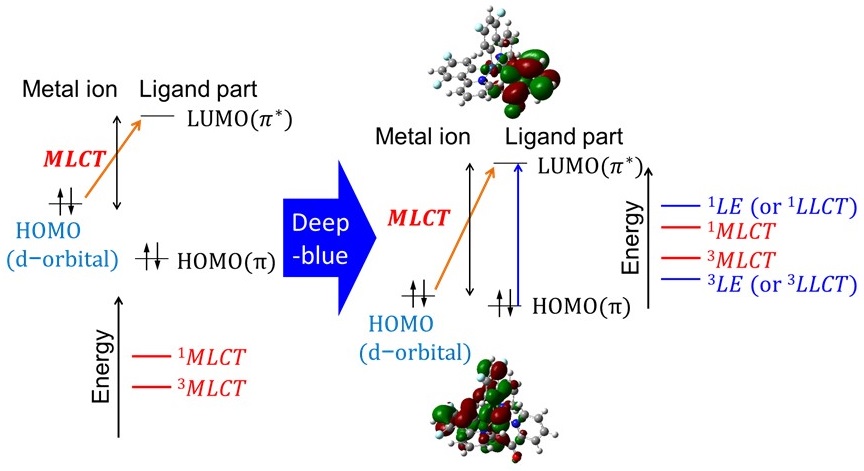
To advance the a posteriori quest for deep-blue phosphorescence, we quantify the degree of metal-to-ligand charge trans-fer (MLCT) upon excitation using density functional theory calculations. We also comprehensively determine how the MLCT nature of a state is correlated with the wavelength of potential phosphorescence through comparing the results of two typical ligand structures, i.e., 2-phenylpyridine and 1-phenyl-3-methylimidazolin-2-ylidene, with those of their derivatives. The MLCT nature can be defined as the difference in the electron density over the central metal ion that contributes to the highest occupied and lowest unoccupied molecular orbitals (HOMO and LUMO, respectively). This study focuses on how the MLCT nature can be characterized upon excitation depending on the admixtures of molecular orbitals and how a stronger MLCT nature is preferable for efficient phosphorescence, while the MLCT nature exhibits a trade-off relationship with the wavelengths of phosphorescence in the blue region. Moreover, we elucidate how iridium(III) complexes are suitable for efficient phosphorescence and discuss a conceptual grand design for transition-metal complexes with deep-blue phosphorescence using the ligand field theory. We examine why strong MLCT nature upon excitation does not necessarily ensure highly efficient phosphorescence, taking into account possible relative energy gaps between the singlet MLCT, triplet locally excited, and d‒d transition states in a transition-metal complex formed after excitation. Nevertheless, the initial MLCT serves as a necessary condition that induces corollary energy-state configurations as well as decay from the excited states. Therefore, quantifying the nature of that MLCT, which is the first process upon excitation, with the possible wavelength of phosphorescence, is a useful parameter for a conceptual grand design for candidate complexes that can efficiently emit deep-blue phosphorescence. This reverse method of analyzing candidate complexes before experimentally formulating and studying them can accelerate the a posteriori quest for deep-blue-emitting materials.